Basic Brain Research into Nerve Development Hints at Mechanisms in Huntington’s
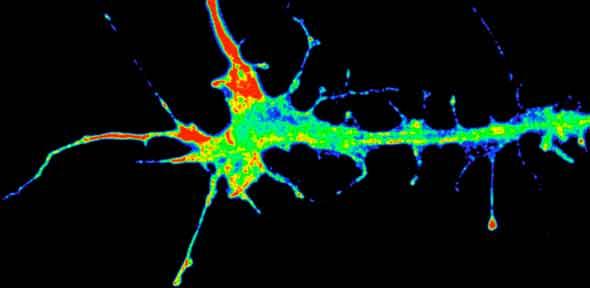
Credit: K-M. Leung
Two scientists at Cambridge University used differing techniques to better understand how brain cells form connections, research that may inspire new ways of rewiring the brain to repair nerves and treat diseases such as Huntington’s.
Professor Christine Holt of the Department of Physiology, Development and Neuroscience, focused on how molecules influence neurons to form connections, known as synapses, in the visual system in response to light signals from the eye’s retina. “It’s an impressive navigational feat,” Dr. Holt said in a news release. “The pathway between the retina and the brain may look homogeneous, but in reality it’s like a patchwork quilt of different molecular domains.”
Dr. Holt studied growth cones, structures found on the top of neurons that seek out partner neurons to form a connection. She focused on the chemical signals that guide growth cones and discovered that these structures can actually make guidance proteins that aid in path-finding. She also found that the same molecules can have different roles depending on the stage of development.
In contrast to Dr. Holt, who studied growing neurons, Dr. Stephen Eglen of the Department of Applied Mathematics and Theoretical Physics utilized mathematical models to understand how nervous system cells might form connections. “You’ve got much more freedom in a theoretical model than you do in an experiment,” Dr. Eglen said. “A common experimental approach is to remove something genetically and see what happens. I think of that a little like taking the battery out of your car. Doing that will tell you that the battery is necessary for the car to function, but it doesn’t really tell you why. It had been thought that if we built a model and took out all of the guidance molecules, there would be no topographic order whatsoever. But instead we found that there is still residual order in how the neurons are wired up, so there must be extra molecules or mechanisms that we don’t know about. What we’re trying to do is to take biology and put it into computers so that we can really test it.”
Using both experiments and computer modeling, researchers could capture the best of two worlds in terms of understanding nervous system development. “In the past 15–20 years, there’s been a revolution in terms of being able to identify the specific molecules that act as guidance receptors or signals, but there’s still so much we don’t yet know, which is why we’re using both theoretical and experimental techniques to answer these questions,” Dr. Holt said.
Taken together, this research could shed light on what happens in nervous system diseases, and possibly identify targets for the treatment of conditions like Huntington’s disease. Said Dr. Holt, “We’ve also found that many of the types of mRNA that are being translated in axons are the same ones that you see in diseases like Huntingdon’s and Parkinson’s, so basic knowledge of this sort is essential for the development of clinical therapies in nerve repair and for understanding these and other neurodegenerative disorders.”
(The image shown is of a growing axon tip exhibiting polarized mRNA translation (red), Credit: K-M. Leung)






