New Brain Imaging Tool Assesses Defective Regions in Neurological Disorders, Huntington’s Disease
Researchers at the University of Pittsburgh and the Carnegie Mellon University recently reported the first non-invasive brain-imaging tool to visualize the brain’s basal ganglia regions, which are thought to be defective in some neurological disorders including Huntington’s disease. The study entitled “In vivo characterization of the connectivity and subcomponents of the human globus pallidus” was published in the journal NeuroImage.
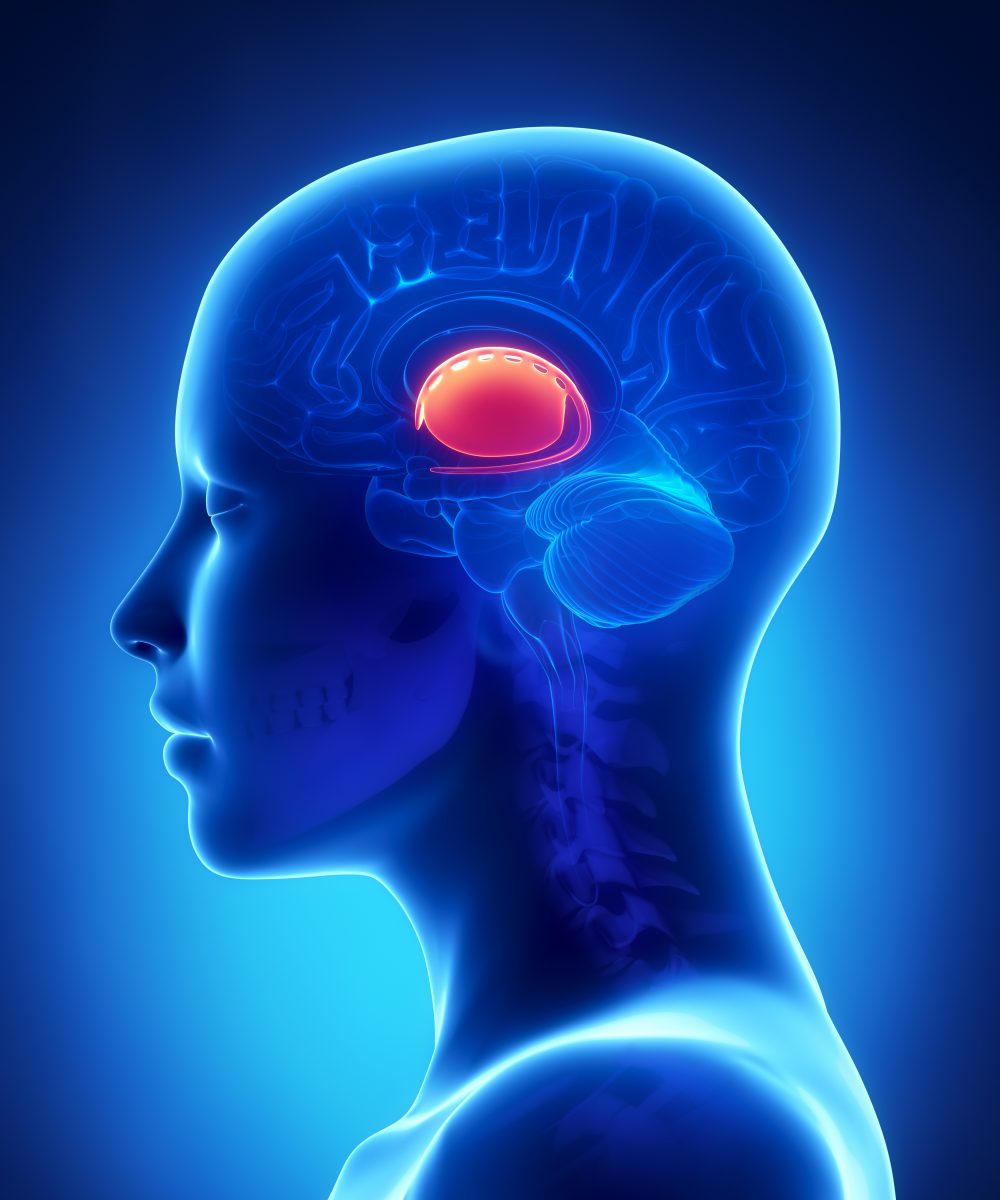
The basal ganglia correspond to a group of structures at the base of the brain. It is responsible for coordinating movement and is composed of four interconnected, deep brain structures. In some disorders, the pathways between the four basal ganglia regions are defective. This is the case in Huntington’s disease, a progressive, neurodegenerative, genetic disorder characterized by the lack of muscle coordination, unsteady gait, mental decline and emotional disturbances. Disease symptoms usually begin in midlife and there is no effective treatment for Huntington’s disease, with one in every 10,000 to 30,000 individuals in the United States suffering from the condition.
Imaging techniques have so far been unable to allow the visualization of the brain structures composing the basal ganglia. “Clinically, it is difficult to see the pathways within the basal ganglia with neuroimaging techniques, like the ever popular MRI [magnetic resonance imaging], because many of the fiber bundles that make up key parts of this circuit are very small and buried within dense cell bodies,” explained the study’s lead author Patrick Beukema in a news release. “For reasons that are not fully understood, the pathways that connect the basal ganglia’s regions are highly susceptible to damage. Because they are important for motor control, this damage can result in substantial motor deficits, so it is highly desirable to discover more about this area,”
Now, for the first time, researchers used a non-invasive brain-imaging tool to visualize these deep brain regions and assess the pathways between them. The team used two types of diffusion imaging, a measurement of the movement of water molecules that allows the creation of a visual representation of the brain’s nerve cells. In total, the brains of 60 healthy adult individuals were analyzed through diffusion spectrum imaging, and 78 through multi-shell imaging.
Researchers found that both imaging techniques detected the small fiber connections in the brain, and analysis of the general patterns of water movement in the basal ganglia made it possible to distinguish one small brain region from the other.
“The pathways that Patrick has been able to visualize are critical to so many functions, yet we haven’t been able to see them in the living human brain before. This opens the door to so many research and clinical opportunities,” said the study’s senior author Dr. Timothy J. Verstynen.
In conclusion, the research team provided new insights into the basal ganglia’s circuitry, which could potentially help develop technologies to monitor disease progression in Huntington’s disease and other neurological disorders.